 Water is arguably the most important chemical compound on our planet. As such, a great deal of scientific research has gone into understanding the structure and dynamics of water as it relates to the biochemical processes in our bodies, environmental science, and even the formation of bubbles. There are many tools in our scientific “toolbox” that can be used to elucidate water’s many mysteries. Arguably the most powerful of those tools is spectroscopy. Spectroscopy comes in many flavors – for example scientists are able to vary the wavelength of light to study different chemical properties of water in order to tease out what is happening on a molecular level.
Water is arguably the most important chemical compound on our planet. As such, a great deal of scientific research has gone into understanding the structure and dynamics of water as it relates to the biochemical processes in our bodies, environmental science, and even the formation of bubbles. There are many tools in our scientific “toolbox” that can be used to elucidate water’s many mysteries. Arguably the most powerful of those tools is spectroscopy. Spectroscopy comes in many flavors – for example scientists are able to vary the wavelength of light to study different chemical properties of water in order to tease out what is happening on a molecular level.
One of the most informative spectroscopic techniques is Raman scattering because it probes the vibrational structure of a molecule with high resolution (Figure 1). Depending on the laser source it is possible to distinguish even the subtlest of changes in the vibrational spectrum, down to fractions of a wavenumber!
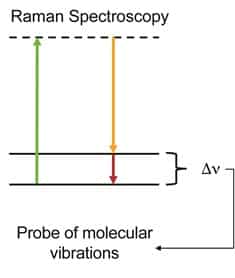
Figure 1. The green arrow shows an excitation to a virtual state via a laser pulse. The yellow arrow depicts the light emitted as the molecule radiatively relaxes to a vibrational level in the electronic ground state. This is what is measured by the detector and the difference between the absorption and emission lines yields the vibrational spectrum of the molecule. Because of its ubiquity in nature, understanding the unique, complex hydrogen bonding network of water has drawn a great deal of interest from the scientific community. In order to tease out the subtle changes in the water vibrational spectrum, high resolution techniques are invaluable, thus bringing Raman spectroscopy to the forefront of the field. Figure 2 depicts the –OH stretch of water probed using a StellarNet 532nm Raman System.
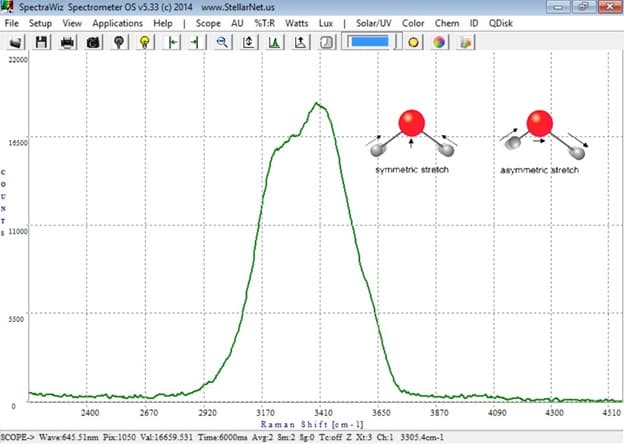
Figure 2. Raman scattering spectrum of pure water. The shoulder at ~3250 cm-1 corresponds to the asymmetric –OH stretch, and the most intense feature at ~3410 cm-1 corresponds to the symmetric –OH stretch. Even more intriguing, there is a small shoulder at ~3630 cm-1 which corresponds to the –OH stretch of a water molecule which is only partially involved in hydrogen bonding – a particularly rare event.
While the Raman scattering technique is characterized by it’s incredibly high resolution, oftentimes information is lost because an experiment is limited by the resolution of the detector. With the advent of StellarNet’s low cost and high resolution spectrometers, more scientists can begin to look at even more subtle changes to the vibrational spectrum than ever before.





