Jiahe Xu, Miharu Koh, Shelley D. Minteer, and Carol Korzeniewski
Published: 13 January 2023 | Electrochemical cells, Electrodes, Ions, Polymers, Redox reactions | Published by the American Chemical Society
Abstract
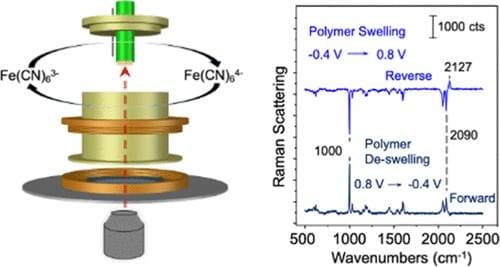
A spectroelectrochemical cell is described that enables confocal Raman microscopy studies of electrode-supported films. The confocal probe volume (∼1 μm3) was treated as a fixed-volume reservoir for the observation of potential-induced changes in chemical composition at microscopic locations within an ∼20 μm thickness layer of a redox polymer cast onto a 3 mm diameter carbon disk electrode. Using a Raman system with high collection efficiency and wavelength reproducibility, spectral subtraction achieved excellent rejection of background interferences, opening opportunities for measuring within micrometer-scale thickness redox films on widely available, low-cost, and conventional carbon disk electrodes. The cell performance and spectral difference technique are demonstrated in experiments that detect transformations of redox-active molecules exchanged into electrode-supported ionomer membranes. The in situ measurements were sensitive to changes in the film oxidation state and swelling/deswelling of the polymer framework in response to the uptake and discharge of charge-compensating electrolyte ions. The studies lay a foundation for confocal Raman microscopy as a quantitative in situ probe of processes within electrode-immobilized redox polymers under development for a range of applications, including electrosynthesis, energy conversion, and chemical sensing.
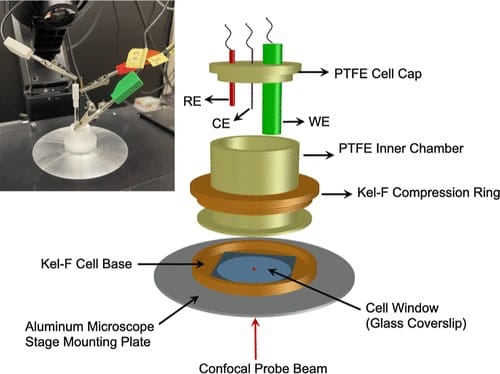
Figure 1. Sketch of the microscope stage mountable cell for confocal Raman microscopy studies of films supported on conventional disk electrodes. A photo of the cell on the microscope stage is shown on the inset. The working, counter, and reference electrodes are labeled WE, CE, and RE, respectively.
… After subtraction of a dark current offset, Raman spectra were corrected for instrument response by ratioing to a recorded spectrum of emission from a standard reference halogen source lamp (SL1-CAL, StellarNet, Tampa, FL, USA) …