Jose L. Trenzado, Cristina Benito, Mert Atilhan, Santiago Aparicio
Published: 24 January 2023 | Department of Physics, University of Las Palmas de Gran Canaria | Department of Chemistry, University of Burgos | Department of Chemical and Paper Engineering, Western Michigan University
Abstract
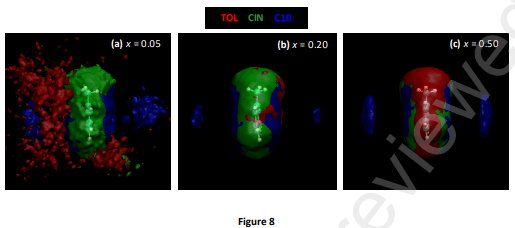
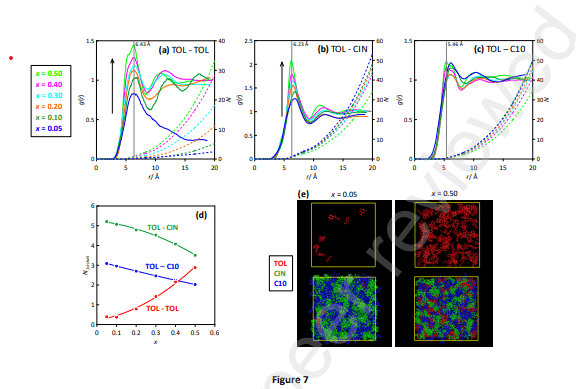
The hydrophobic deep eutectic solvent formed by the equimolar combination of natural compounds cineole and decanoic acid is studied as an archetypical compound for aromatics (toluene) solubilization/extraction purposes. The physicochemical properties of toluene solutions in the solvents are studied as a function of composition and temperature. The nature of intermolecular forces between toluene and solvent molecules is analyzed using a minimal clusters approach through quantum chemistry methods. Classical molecular dynamics simulation on toluene solution allowed us to infer nanoscopic behavior and structuring of the toluene mixtures using static and dynamic approaches. Likewise, the possible biological effects of the considered solvent are studied considering its effect on lipid bilayers as a model of plasma membranes using coarse-grained molecular dynamics methods. The reported results provide a multiscale combined experimental and computational characterization of hydrophobic Deep Eutectics as suitable solvents for aromatic hydrocarbons.
Keywords: hydrophobic deep eutectic solvents; aromatics recovery; molecular modeling; lipid bilayers.
“Deep Eutectic Solvents (DESs) [1,2] are composed of two or more compounds, which upon mixing at suitable ratios, lead to a significant decrease in melting point in comparison with those of pure components [3,4]. Up to five classes are defined for DESs [5], being the most relevant for practical purposes are those formed by two types of compounds, namely Hydrogen Bond Donor (HBA) and Hydrogen Bond Acceptor (HBA), which develop strong hydrogen bonds being the molecular level reason of the melting point decreases [6,7].”
… Raman spectra of liquid samples at ambient temperature (293 +/- 1 K) for a 785 nm laser excitation wavelength were collected using a StellarNet-HR-TEC-785 spectrometer (4 cm-1 resolution), including an enhanced CCD array detector …